A Solar Cell That Does Double Duty for Renewable Energy
Researchers develop an artificial photosynthesis system that generates hydrogen fuel and electricity at the same time.
In the quest for abundant, renewable alternatives to fossil fuels, scientists have sought to harvest the sun’s energy through “water splitting,” an artificial photosynthesis technique that uses sunlight to generate hydrogen fuel from water. But water-splitting devices have yet to live up to their potential because there still isn’t a design for materials with the right mix of optical, electronic, and chemical properties needed for them to work efficiently.
Now researchers at the U.S. Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab) and the Joint Center for Artificial Photosynthesis (JCAP), a DOE Energy Innovation Hub, have come up with a new recipe for renewable fuels that could bypass the limitations in current materials: an artificial photosynthesis device called a “hybrid photoelectrochemical and voltaic (HPEV) cell” that turns sunlight and water into not just one, but two types of energy – hydrogen fuel and electricity. The paper describing this work was published on Oct. 29 in Nature Materials.
Finding a way out for electrons
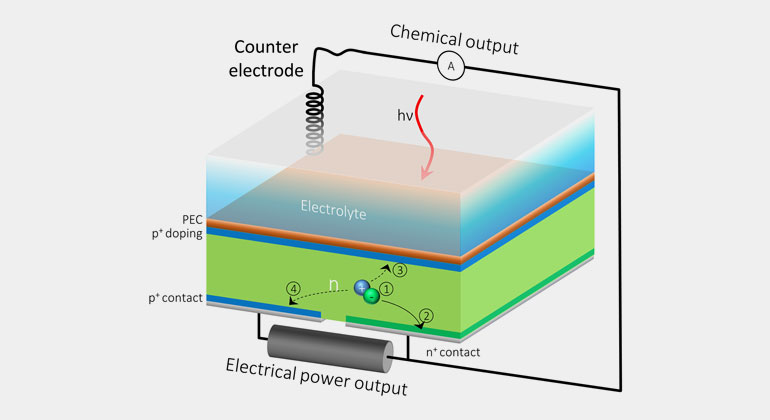
Most water-splitting devices are made of a stack of light-absorbing materials. Depending on its makeup, each layer absorbs different parts or “wavelengths” of the solar spectrum, ranging from less-energetic wavelengths of infrared light to more-energetic wavelengths of visible or ultraviolet light.
When each layer absorbs light it builds an electrical voltage. These individual voltages combine into one voltage large enough to split water into oxygen and hydrogen fuel. But according to Gideon Segev, a postdoctoral researcher at JCAP in Berkeley Lab’s Chemical Sciences Division and the study’s lead author, the problem with this configuration is that even though silicon solar cells can generate electricity very close to their limit, their high-performance potential is compromised when they are part of a water-splitting device.
The current passing through the device is limited by other materials in the stack that don’t perform as well as silicon, and as a result, the system produces much less current than it could – and the less current it generates, the less solar fuel it can produce.
“It’s like always running a car in first gear,” said Segev. “This is energy that you could harvest, but because silicon isn’t acting at its maximum power point, most of the excited electrons in the silicon have nowhere to go, so they lose their energy before they are utilized to do useful work.”
Getting out of first gear
Lead author Gideon Segev and co-author Jeffrey W. Beeman are both JCAP researchers in Berkeley Lab’s Chemical Sciences Division. (Credit, left-right: Marilyn Chung/Berkeley Lab, Jeffrey W. Beeman/Berkeley Lab).
So Segev and his co-authors – Jeffrey W. Beeman, a JCAP researcher in Berkeley Lab’s Chemical Sciences Division, and former Berkeley Lab and JCAP researchers Jeffery Greenblatt, who now heads the Bay Area-based technology consultancy Emerging Futures LLC, and Ian Sharp, now a professor of experimental semiconductor physics at the Technical University of Munich in Germany – proposed a surprisingly simple solution to a complex problem.
“We thought, ‘What if we just let the electrons out?’” said Segev.